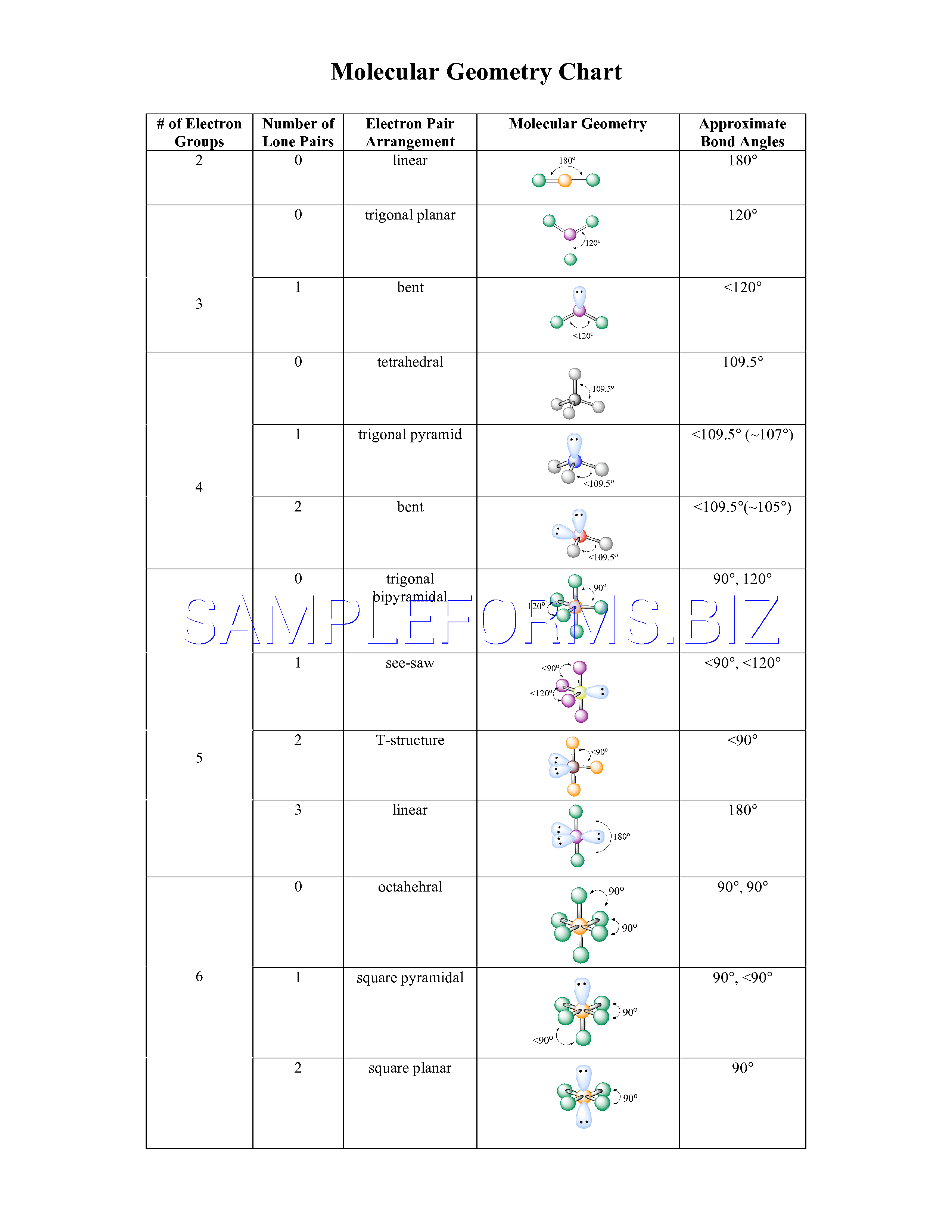
 We have s,p,d,f atomic orbitals ( AOs ).Įlectrons can be explained via their wave functions in quantum mechanics. An orbital, on the other hand, is defined to be the probability of the presence of an electron in any given regional space.Ītomic orbitals are of several shapes like spherical, dumb-bell, clover-leaf, or doughnut to name a few of the complex varieties. In chemistry, an orbit is a fixed path around the nucleus in which electrons tend to move or revolve. The bond angle is around 103 degrees (due to the repulsion of the lone pair). According to the above chart, we have a bent molecule structure. The VSEPR notation for the OF2 molecule is AX2E2. of lone pairs of electrons of the central atom, ‘X = 2. of surrounding atoms, ‘n’ = 2 ( Fluorine )Į: no. Let us look at this VSEPR chart showing AXN notations:Īccording to AXnEx notation of VSEPR theory, The VSEPR model tends to reduce or minimize the repulsive forces and balance the stability of any given molecule, in this case, OF2. Like charged electrons create a negatively charged cloud surrounding the atomic nuclei and repulsion is created. Our next step is to use Valence Shell Electron Pair Repulsion Theory (VSEPR) to find the exact molecular geometry along with the bond angles. Via Lewis Structure, we have realized the type of bond formed and the number of lone or unbonded pairs of valence electrons present in an OF2 molecule. Now, we are going to decipher the 3D molecular shape. We have already found the 2D Lewis Structure diagram of the Oxygen Difluoride molecule. Here, oxygen forms a single bond with each of the fluorine atoms (O-F). Since the elements are present in their least possible formal charge values, we have achieved our suitable Lewis Structure configuration. The same goes for the central oxygen atom.īefore we proceed to confirm any sketch to be the perfect Lewis Structure for a given molecule or ionic structure, we need to check the formal charge values.įor Oxygen: Formal Charge = 6 – 0.5*4 – 4 = 6 – 2 – 4 = 0.įor each Fluorine atom: Formal Charge = 7 – 0.5*2 – 6 = 7 – 1 -6 = 0. Here, if we look at the octet rule, both the fluorine atoms have achieved a valency of eight. This is known as octet fulfillment since the elements want to achieve eight electrons in their valence shells.įor example, Carbon tends to attain a Neon configuration.Įxception: Hydrogen tends to achieve Helium configuration, hence only two electrons in the outer shell. The main group elements ( group 1 to group 17 ) of the periodic table have a tendency to attain the octet configuration of the noble gas elements present in group 18 of the same period.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
